New research available on diagnostic tests for COVID-19
The knowledge of diagnostic tests to detect SARS-CoV-2 infection is still evolving. A recent article describes how to interpret the two types of diagnostic tests commonly used, the RT-PCR and IgM/IgG tests.
There is further clarification on RT-PCR and antibody tests
The knowledge of diagnostic tests to detect SARS-CoV-2 infection is still evolving. A recent article describes how to interpret the two types of diagnostic tests commonly used for SARS-CoV-2 infections - RT-PCR and IgM/IgG tests (ELISA) - and how results may vary over time.
Made in cooperation with our partners from esanum.it
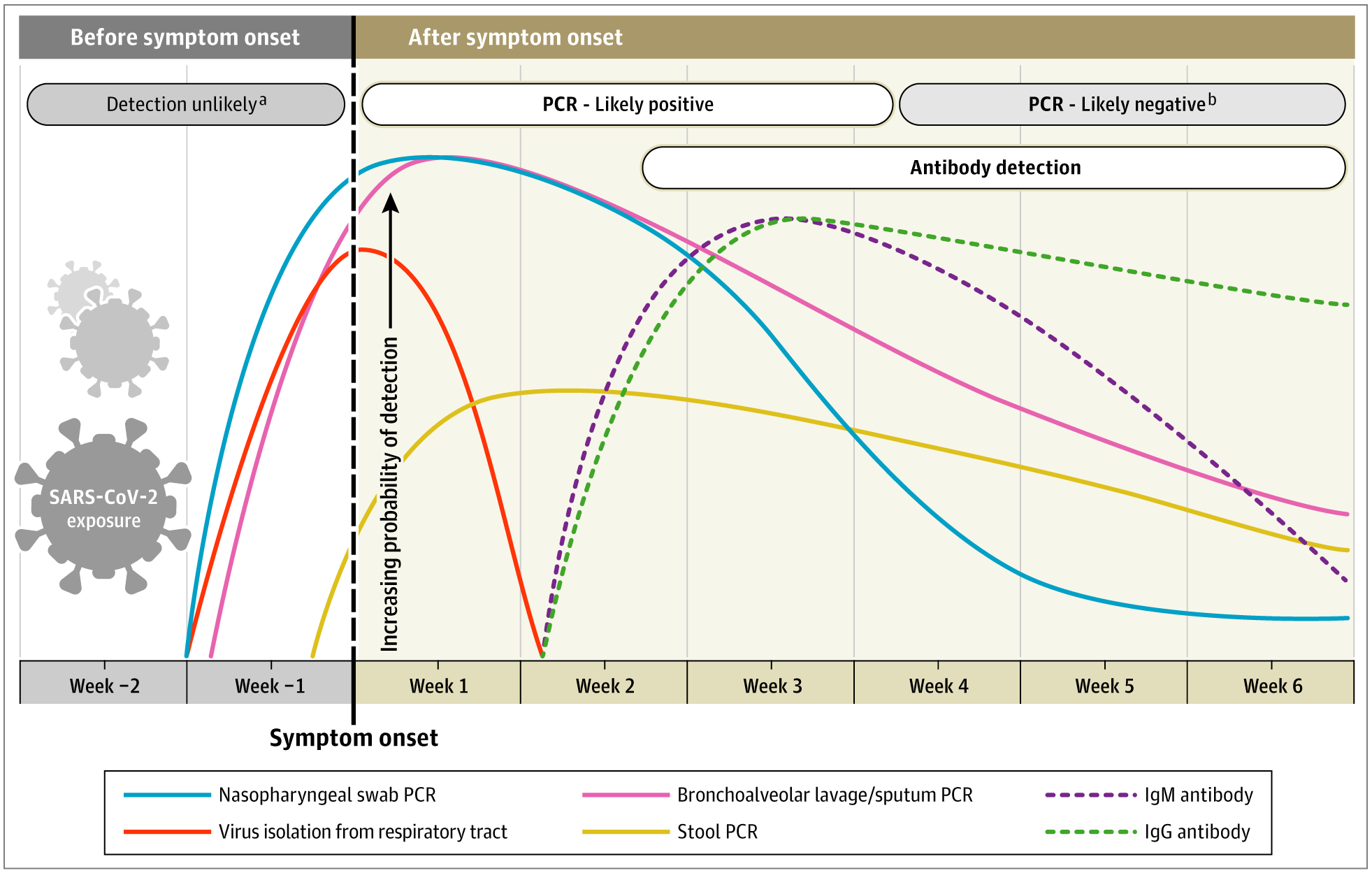
An article published in JAMA (The Journal of the American Medical Association) reports a timeline of diagnostic markers for the detection of COVID-19 based on currently available data. Most of the available data refer to adult immunocompetent populations. The time course of RT-PCR positivity and seroconversion may vary in children and other groups.
Detection of viral RNA via RT-PCR
So far, the most commonly used and reliable test for the diagnosis of COVID-19 has been the RT-PCR test performed using nasopharyngeal and oropharyngeal swabs. Most kits on the market target one or more genes from the various viral components.
In most individuals with symptomatic SARS-CoV-2 infection, the viral RNA in the nasopharyngeal swab becomes detectable from the first day of onset of symptoms and peaks in the first week. This test positivity begins to decrease within the third week and then becomes undetectable. However, RT-PCR positivity may persist more than 3 weeks after the onset of the disease, when most mild cases will give a negative result. It should be noted that a positive RT-PCR result reflects only the detection of viral RNA and does not necessarily indicate the presence of viable viruses. In some cases, viral RNA has been detected by RT-PCR even beyond the 6th week after the first positive swab.
The timeline of RT-PCR positivity differs in samples other than a nasopharyngeal swab. Positivity decreases more slowly in sputum and may still be positive after nasopharyngeal swabs are already negative.
Detection of SARS-CoV-2 antibodies
COVID-19 infection can also be detected indirectly by measuring the host's immune response to SARS-CoV-2 infection. Serological diagnosis, besides having a diagnostic role, is also becoming an important tool to understand the extent of COVID-19 in the community and to identify individuals who are immune and potentially "protected" from infection. The most sensitive and early serological marker is total antibodies, whose levels begin to increase from the second week from the onset of symptoms.
According to current knowledge, seroconversion of the immunoglobulins IgM and IgG occurs between the third and fourth weeks of the onset of clinical disease. Subsequently, the IgMs begin to decrease, reach lower levels by the fifth week and almost disappear by the seventh week. On the other hand, IgG persists beyond 7 weeks.
In a study with 140 patients, the combined sensitivity of RT-PCR and IgM ELISA to nucleocapsid antigen (NC) was 98.6% compared to 51.9% in the single RT-PCR test. ELISA-based IgM and IgG antibody tests have a specificity greater than 95% for the diagnosis of COVID-19. Analysis of serum samples in combination with an initial buffer test and a second after two weeks can further increase diagnostic accuracy.
Rapid tests for antibody detection have been widely developed and marketed and are of variable quality. Many manufacturers do not reveal the nature of the antigens used. These tests are of a purely qualitative nature and can only indicate the presence or absence of SARS-CoV-2 antibodies. Many questions remain, particularly with regards to how long potential immunity lasts in individuals, both asymptomatic and symptomatic, who are infected with SARS-CoV-2.
Source:
Sethuraman N, Jeremiah SS, Ryo A. Interpreting Diagnostic Tests for SARS-CoV-2 [published online ahead of print, 2020 May 6]. JAMA. 2020;10.1001/jama.2020.8259. DOI:10.1001/jama.2020.8259